Abstract
This study evaluated the Lake Louise Score (LLS) alongside key physiological parameters—arterial oxygen saturation (SpO2) and heart rate—across various altitudes in the Langtang region. Data were gathered from participants at Syabrubesi, Langtang Village, Kyanjin Gompa, Tsergo Ri, and Lama Hotel, with a focus on assessing the impact of gender and smoking habits on these parameters. Descriptive statistics and paired sample tests were employed for the analysis.
The participant pool was predominantly male (84.8%), with females constituting 15.2%. Smokers made up 51.5% of the sample, while non-smokers accounted for 48.5%. SpO2 levels demonstrated a decline with increasing altitude in both smokers and non-smokers, with smokers consistently showing lower SpO2 levels. The variability in SpO2, as indicated by standard deviation, increased at higher altitudes.
Heart rate trends indicated an elevation in heart rate corresponding to higher altitudes. Smokers exhibited higher heart rates than non-smokers, and heart rate variability also increased at elevated altitudes.
Gender-based differences were notable, with females generally maintaining higher SpO2 levels compared to males at equivalent altitudes, possibly due to hormonal influences. Heart rate responses to altitude varied, with some evidence pointing to elevated heart rates in females, although findings on gender differences were inconsistent.
The results of this study offer critical insights into the interaction between the Lake Louise Score and physiological responses to altitude, influenced by gender and smoking habits. These findings underscore the necessity for further research to elucidate the underlying mechanisms and individual variability in these responses, aiming to enhance health management and acclimatization strategies for high-altitude exposure.
Keywords: Lake Louise Score, altitude, physiological parameters, arterial oxygen saturation, heart rate, gender differences, smoking habits
Introduction
Every year, a large number of sojourners can visit highland or moderate-altitude areas without any special training or acclimatization; or adaptation to the new environment. This pattern has had a greater impact on highland trekking accidents (Saito et al., 20051). Acute Mountain Sickness (AMS), High-altitude Cerebral Edema (HACE), and High-altitude Pulmonary Edema (HAPE), generally known as altitude sickness. Moghaddasi, 20192; wall and Paudel, 20213; Basnyat et al., 20004 Karine et al., 20105 reported shows that AMS, HAPE and HACE are all encountered by travelers who are more than 1900 asl, 2000 asl, 2500 asl, and 2700 asl. The recorded frequency various greatly, from 9% in the Swiss Alps at 2850 asl to over 50% in the Nepal Himalaya at 4200 asl (karine et al., 20086). According to the literature, elevations above 1500 asl increase the risk of getting altitude sickness, however, there may be asymptomatic. High altitude sickness is common among these travelers at this altitude, but it becomes a health issue or a life challenge, and it can be fatal.
The most common altitude sickness refereed as AMS which is experienced or encountered by newcomers to highlands. They experience discomfort with their body function, which manifests as a syndrome non-specific such as shortness of breath, headache, sleep apnea, insomnia, rapid pulse, nausea or vomiting, fatigue, dizziness or light-headache (Luks et al., 20177). The partial pressure of oxygen decreases as the barometric pressure decrease at high altitudes. The hypoxia causes a series of physiological responses that in most cases aid in the individual’s adaption to low oxygen levels and high altitude. In some cases, however, maladaptive response occurs, resulting in high altitude illness (Moghaddasi, 20198).
Literature review
With the advancement of new technology, newcomers or sojourners can visit high altitude or moderate areas without any special altitude training or acclimatization which has had a significant impact on highland trekking accidents (Saito et al., 20059). When ascending to a high altitude, the body experiences hypoxic hypoxia and low atmospheric pressure, which takes some time for the body to adjust (Eter et al., 200110). If the acclimatization or body has not adjusted to this highland altitude or fails to the adaption process because of the rapid ascend and without any special altitude training and preparation rate off the vulnerability of the visitors, one or more of three altitudes related sickness may result from Acute Mountain Sickness (AMS), high altitude Cerebral Edema (HACE), and High-Altitude Pulmonary Edema (HAPE). Among the three altitude related illnesses AMS is the most common problem affecting 25 % of those who ascend to an altitude of 1850 to 2750 asl (Honigman et al., 1993), 42 % at an altitude of 3000 asl (Eter et al., 200111) and at the altitude of 5984 asl of mountain Kilimanjaro 75% has reported (H. Karinen et al., 200812). Thus, these studies show the incidence of high-altitude Sickness after the first ascent. (Hackett et al., 197613) studies shows that the overall incidence of an illness is 53%. In another study by Meier et al., 201714), the elevation above 3500 asl more than 25 % and more than 50 % above 6000 asl may occur AMS less than 1% of people have seen or may progress non-specific or asymptomatic symptoms of AMS to life-threatening high-altitude cerebral edema (HACE).
The cause of altitude sickness is a matter of oxygen physiology. At sea level, the oxygen concentration is approximately 21%, and the barometric pressure averages 760 mmHg. As altitude increase, the oxygen concentration remains constant, but the number of oxygen molecules per breath decreases. At 3,658 asl, the barometric pressure is only 483 mmHg, so there are roughly 40% fewer oxygen molecules per breath (Richalet et al., 201115). To oxygenate the body effectively, the breathing rate (even while at rest) has to increase. This extra ventilation increases the oxygen content in the blood, but not to sea level oxygen concentrations. Whereas the required amount of oxygen for activity is the same. However, the body must adjust to having less oxygen.
Wall and Paudel, 202116 report suggested, high altitude and lower air pressure cause fluid to leak from the capillaries which can cause fluid buildup in both lungs and the brain. Continuing to higher altitudes without proper acclimatization can lead to potentially serious, even life- threatening illnesses. High altitude sickness is caused by the body retaining excess bodily fluids that accumulated in a certain part of the body as a result of exposure to the low pressure of oxygen and build-up of the red blood cells which is not sufficient transporting the required or excess fluid in the whole body or system.
High Altitude
In the field of high-altitude medicine, high altitude refers to terrestrial elevations greater than 1500 asl (4921 ft) (Gallagher and Hackett, 200417) and is commonly classified into three categories: High altitude, very high altitude and extreme altitude (Table 1). According to (Gallagher and Hackett, 200418) studies, the environment at high elevations become more hostile as one ascends, necessitating increased levels of behavioral and physiological adaption to maintain function and long-term survival. Acclimatization is the process through which an individual body adjust to the lower ambient oxygen pressure at a high altitude. Low humidity, reduced temperature and increased ultraviolet radiation are some additional environmental challenges associated with high altitude. Medical issues may arise from these conditions.
Table 1: Definition of High, Very High, and extreme altitude
| Altitude | Mean above sea level (masl) | Feet |
| High altitude | 1,500-3,500 | 5,000-11,500 |
| Very high altitude | 3,500-5,500 | 11,500-18,000 |
| Extreme altitude | Above 5,500 | Above 18,000 |
Generally, the oxygen concentration at sea level is approximately 21 % and the barometric pressure is about 760 mmHg. Barometric pressure and inspired oxygen are distinct at various altitudes (Table 2). The oxygen concentration at high altitudes same at sea level but the number of Oxygen in each breath is decreased (Wall and Paudel, 202120). At the altitude of 4000 asl only 475 mmHg barometric pressure which is roughly 40% fewer oxygen molecules in each breath. As a result, the breathing rate increase even at rest which decreases inspired oxygen at hypobaric hypoxia is the major factor for the development of altitude illness (Hevroni et al., 201521).
Table 2: Barometric Pressure and Inspired Po2 at various Altitudes
| Altitude (masl/feet) | Barometric Pressure (mmHg) | Inspired PO2, mmHg (% of sea level) |
| 0(0) | 760 | 149(100) |
| 1000(3281) | 679 | 132(89) |
| 2000(6562) | 604 | 117(79) |
| 3000(9843) | 537 | 103(69) |
| 4000(13123) | 475 | 90(60) |
| 5000(16404) | 420 | 78(52) |
| 8848.86(29031.6929) | 253 | 43(29) |
Lowlands residents who make their journey at high altitudes with different purposes such as recreational, religious, economic, mountaineering, research and military are at high risk. They are experiencing some degree of altitude illness. categories of high altitude with the physiological effects at various elevations and the normal ranges of arterial oxygen saturation SPo2 are shown in Table 3.
Table 3: Categories of high altitude and their physiologic effects
| High Altitude | masl | SPO2 normal range (%) | Physiology affects |
| High altitude | 1,500-3,500 | Minar impairment in atrial saturation | High-altitudes illness common with an abrupt ascent to above 2500 masl(8202 feet) Decreased exercise performance and increased ventilation. |
| Very high altitude | 3,500-5,500 | 75-85 | The most common range for severe high-altitude illness abrupt ascent may be dangerous a period of acclimatization. |
| Extreme altitude | Above 5,500 | 58-85 | Progressive deterioration of physiologic function eventually outstrips acclimatization above the highest permanent human habitation abrupt ascent almost always precipitates severe high-altitude illness. |
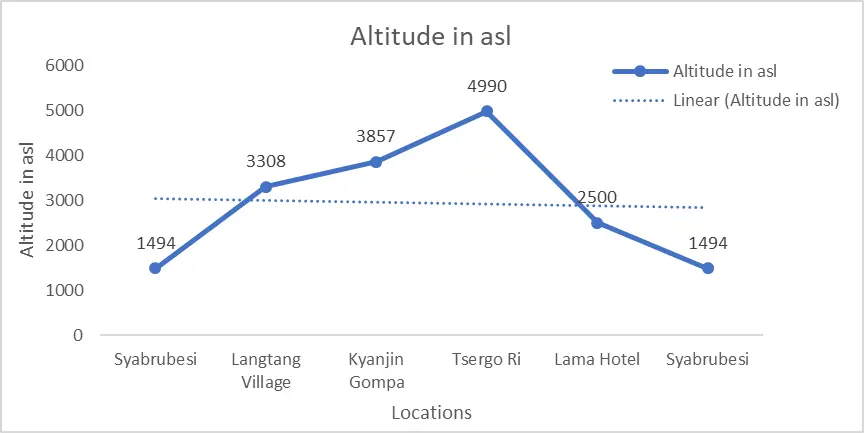
As gained elevation, individual experience difficult to breath. He/she used to inhale oxygen as required to feel comfort or easy to perform activities. As we gained elevation, the amount of air in atmosphere is same as at sea level 21%, but the thin air, the amount of oxygen in each breath is decreased. As a result, individual experience to uncomfortable to breath. The practical approached practiced by mountaineer or mountain guide, which has shown in figure 1.

Image I: Approximate body oxygen saturation (SpO₂) percentages at various altitudes, ranging from 1,350 meters to 5,500 meters. This data highlights the physiological adaptation of the human body to decreasing oxygen levels with increasing elevation.
Image II:Relationship between body oxygen saturation (SpO₂) percentages and oxygen availability at varying altitudes, from sea level to 8,848.86 meters. This chart emphasizes the decreasing oxygen availability as altitude increases, crucial for understanding the risks of altitude sickness in mountaineering.
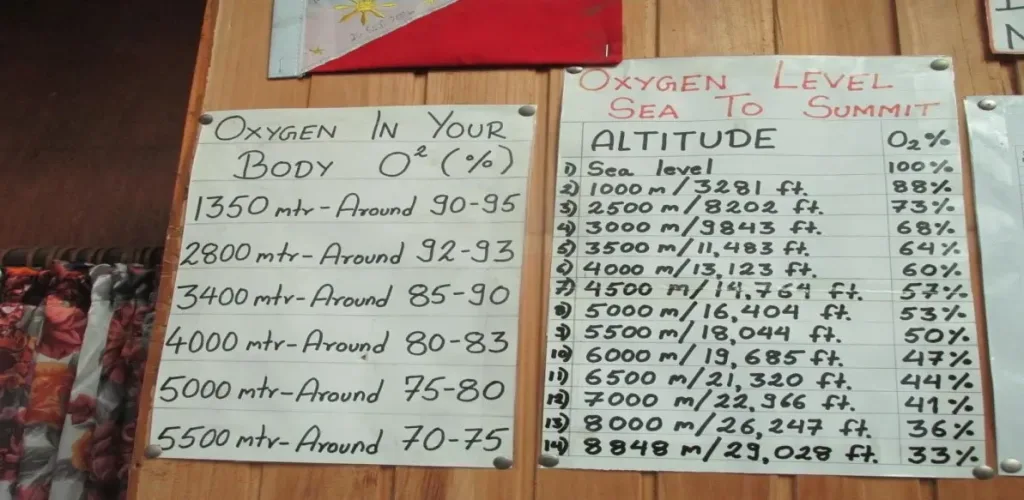
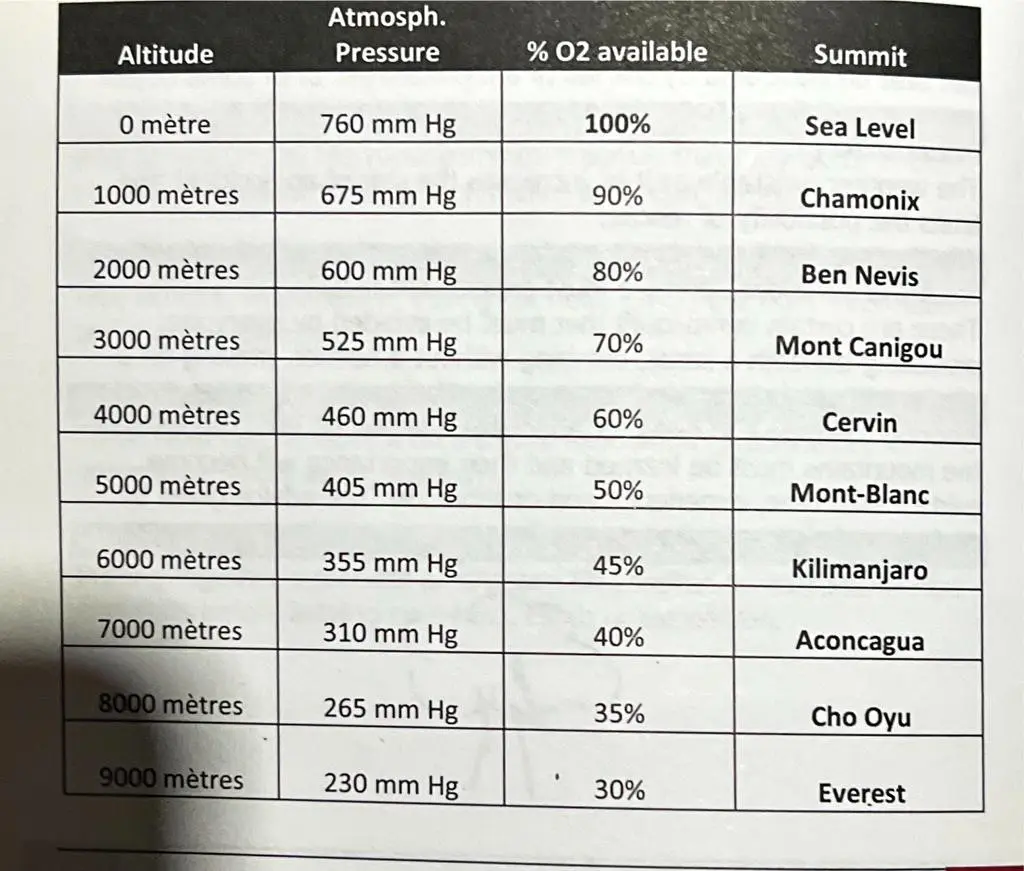
Image III: Correlation of altitude with atmospheric pressure, oxygen availability percentages, and notable mountain summits, ranging from sea level to Mount Everest. This table serves as a resource for climbers to assess potential oxygen needs and acclimatization strategies at high elevations.
Figure 1: Normal Spo2 level as practiced
High Altitude illness
The term “High altitude illness” refers to the cerebral and pulmonary syndromes that can occur in unacclimatized individual shortly ascending to high altitude (Eter et al., 200122). In another study by Kurtzman & Caruso (201823) defined high altitude illness as travel to high altitude regions and mountainous area has become increasingly popular for tourism, recreation, adventure activities, and sometimes rescue missions. Hackett and Roach estimate 30 million people travel to mountainous regions of western united states annually. Tens of thousands of travelers visit high-altitude destinations throughout the world each year among them most of the visitors have experienced symptoms of high-altitude illness. Thus, it is a collective term which includes Acute Mountain Sickness (AMS), High altitude cerebral edema (HACE), High altitude pulmonary edema (HAPE) and other several forms such as high-altitude retinopathy, retinal hemorrhage, high altitude syncope, cerebral venous thrombosis and cortical blindness.
Acute Mountain Sickness
Acute Mountain Sickness (AMS) is the most common non-specific syndrome that arise in non-acclimatized visitors who have recently arrived at a high altitude above 2500 asl the presence of headache with the following symptoms: gastrointestinal symptoms (anorexia, nausea, vomiting), insomnia, dizziness, and lassitude or fatigue (Roach et al., 201824). Similarly, the study of Moghaddasi, 201925; wall and Paudel, 202126; Basnyat et al., 200027 and Karinen et al., 200428 demonstrated visitor encountered AMS at the elevation of continuously 1900 asl, 2000 asl, 2500 asl, and 2700 asl. The symptoms appear within six hours of arriving at a high altitude and generally disappear within three days if the visitors are allowed to rest or do not gain additional altitude. Prevalence of AMS at different altitude because the lack of standardization in altitude, ascent rate, outcome, and population. How high and how quickly visitors ascend to high altitude determines the significant risk factors of AMS.
AMS is a clinical diagnosis based on symptoms in the context of ascending to high altitude. Various studies studied several questionnaires-based diagnosis tools that can be used to diagnosis AMS. The Lake Louise Questionnaire Score (LLQS) is a commonly used tool for the diagnosing AMS (Table 4). Some authors have suggested a higher cutoff for diagnosis AMS (Maggiorini et al., 200129), but the consensus committee believes that by eliminating the sleep question, more people with true AMS will be identified at the threshold of three points, including headache. Sufficient research is lacking to divide the score into severity rankings. For those who wish to do so, they suggested mild AMS as 3-5 points, moderate AMS as 6-9 points, and severe AMS as 10-12 Points (Roach et al.,201830).
Table 4: AMS diagnosis rank
| AMS Points | Ranking |
| 3-5 | Mild |
| 6-9 | Moderate |
| 10-12 | Severe |
Diagnostic Criteria for Acute Mountain Sickness
A short self-report questionnaire makes up the Lake Louise score system. It is designed for AMS researchers to use. It is not designed for use by professionals, qualified outdoor guides, or laypeople in the diagnosis or treatment of AMS. A recent elevation gain, at least several hours at the new elevation, a headache, and at least one of the following symptoms—gastrointestinal upset (anorexia, nausea, or vomiting), fatigue or weakness, dizziness or lightheadedness, and difficulty sleeping required for an AMS diagnosis. AMS is defined as a score of three or more on the AMS Self-report questionnaire alone (Roach et al.,200031). This portion of the scoring system is required in self-report questionnaires and should be reported as a separate score. Each of the five questions below is followed by a 0 to 3 response rating. In some studies, the question “Difficulty sleeping” is irrelevant (for example, rapid one-day ascent) and can be skipped (The Lake Louise AMS Scoring System Consensus Committee). The AMS Self-report score is calculated by adding the sum of the answers to these questions (Table 5).
Table 5: Lake Louise AMS Scoring System
| Physical conditions | Score |
| Headache | 0 -No headache 1-Mild headache 2-Moderate headache 3- Severe headache, incapacitating |
| Gastrointestinal Symptoms | 0- No gastrointestinal symptoms 1- Poor appetite or nausea 2-Moderate nausea or vomiting 3-Severe nausea & vomiting, incapacitating |
| Fatigue and or Weakness | 0-Not tired or weak 1-Mild fatigue/weakness 2-Moderate fatigue/weakness 3-Severe fatigue/weakness, incapacitating |
| Dizziness/Light-headedness | 0 -Not dizzy 1-Mild dizziness 2-Moderate dizziness 3-Severe dizziness, incapacitating |
| Difficulty Sleeping | 0-Slept as well as usual 1-Did not sleep as well as usual 2-Woke many times, poor night’s sleep 3-Could not sleep at all |
Source: The Lake Louise AMS Scoring System Consensus Committee
Altitude Acclimatization
Acclimatization is the process of adapting to a new place or climate. It is a physiological adaption which takes time typically takes 2-5 days (Moghaddasi, 201932) that depend on the individual body physiology which involve gradual, long-term responses of an organism to change its environment.
Individuals at low elevations (2400 m) and those who ascend to very high altitudes (>3500 m) or extreme altitudes (>5500 m) are susceptible to high altitude illness (Gallagher and Hackett, 200433), which can result in significant degradation of both physical and cognitive work performance (Fulco et al., 199834).
Altitude acclimatization refers to a set of physiological adjustments that enable the body to cope with the reduced levels of oxygen in the atmosphere. It is considered to be the most effective approach to prevent acute mountain sickness (AMS) (Society & Forgey, 199535) and to optimize physical and mental performance at high altitudes, according to studies conducted by Fulco et al. (200036) and (Banderet & Shukitt-Hale, 200237), to induce altitude acclimatization, it is advisable to adopt a gradual or staged ascent, with a maximum first-night sleeping altitude of 2400 m and a daily elevation gain between 300 and 600 m, as recommended by Society & Forgey (199538).
The acclimatization process to altitude varies depending on several factors, including the altitude, rate of ascent, individual’s physiology, and previous altitude experience. Generally, two key adaptations process are occurred that are increased ventilations and decreased total body water, resulting in a reduced plasma volume (i.e., hem concentration) which stimulates development of altitude acclimatization (Honigman et al., 199339).
Ventilatory acclimatization to high altitude is characterized by a gradual increase in ventilation, arterial oxygen partial pressure, and oxygen saturation (Sao2), as well as a decrease in arterial carbon dioxide partial pressure and normalization of arterial pH within the first 5 to 9 days of exposure to high altitude (Bisgard and Forster, 199640). This increase in ventilation is accompanied by hemoconcentration, which leads to an increase in the oxygen-carrying capacity of the blood (Durkot et al., 199641). After approximately 7 days at high altitude, arterial oxygen content is nearly normalized due to these physiological changes (Sawka et al., 200042).
It is important to note that acclimatization to one altitude does not necessarily confer complete acclimatization to a higher altitude. The time required for an individual to become fully acclimatized depends on their physiological characteristics and the severity of the hypoxic challenge posed by the altitude. Acetazolamide can be used to accelerate the process of ventilatory acclimatization (Rs & Sm, 196743).
Individuals who have not recently acclimatized to high altitude (i.e., within the past month) require the most physiological compensation and therefore take the longest to acclimatize. In contrast, individuals who reside at moderate or high altitudes may acclimatize more quickly to a higher altitude (Beidleman et al., 200444).
Prevention of High- altitude Illness
Widely advocated and most practice to effectively prevent AMS is slow ascent and allowing time to acclimatization (Eter et al., 200145). Available studies suggested that between 300 to 600 masl is the actual rate of ascent with a maximum first-night sleeping a day higher than elevation above 2400 masl which is recommended by Society, W. M., & Forgey, W. W. (199546). AMS incidence of 50-75% when ascending more than 500 masl a day at higher than 4000 masl. The wilderness medicine society developed the guideline that ascend should not be higher than 500 masl a day at altitudes of more than 300 masl, and one day rest at every 3-4 days should be included (Moghaddasi, 201947). Preventing high altitude sickness by resting the first 48 hours or 2 days after arriving at a place of high altitude is advisable (Roach et al., 200048). Some hikers are more susceptible to high altitude illness (Table 6).
Table 5: Trekkers at risk of high-altitude illness
| -Anyone ascending to 2800 masl in one day -Anyone with prior history of acute mountain sickness ascending higher than 2800 masl -Anyone ascending higher than 500 masl/day at altitudes higher than 3000 masl -Anyone with a prior history of severe acute mountain sickness or high- altitude cerebral edema or pulmonary edema |
(Source: Moghaddasi, 201949)
Oxygen Saturation
Oxygen saturation refers to the measure of how much oxygen is in blood, organs and tissue need oxygen to work. For healthy adults, a normal oxygen saturation level is between 95%-100 % oxygen at sea level (<1500 masl). Blood getting saturated with the presence of oxygen (O2), In the healthy adults Lungs millions of microscopic air sac called alveoli exchange oxygen and carbon dioxide molecules to and from the bloodstream. Oxygen saturation is measured in one of the two ways: an arterial blood gas test (ABG or Sao2) and pulse oximetry (SPO2). SaO2 is a critical parameter in the assessment of respiratory function and is routinely measured in clinical practice. SpO2 (Peripheral Capillary Oxygen Saturation) Pulse oximeters are non-invasive medical devices that measure the oxygen saturation in arterial blood by detecting the absorption of light at two wavelengths (red and infrared) through a pulsating vascular bed, usually in the fingertip or earlobe. The ratio of oxygenated hemoglobin (HbO2) to deoxygenated hemoglobin (Hb) determines the SpO2 value, which is expressed as a percentage.
However, it is important to note that pulse oximeters can be affected by various factors, such as motion artifacts, poor peripheral perfusion, skin pigmentation, and ambient light interference. Additionally, altitude-induced hypoxemia can also lead to measurement errors in SpO2 readings. At higher elevations, the atmospheric pressure and oxygen concentration decrease, leading to lower oxygen saturation levels in arterial blood. As a result, pulse oximeters can overestimate the SpO2 readings at higher elevations, leading to potential clinical misinterpretation and inappropriate oxygen therapy.
In a study by (Robergs & Landwehr, 200250), they found that pulse oximeters tended to overestimate SpO2 readings at elevations above 1600 m. They suggested that an altitude correction factor be applied to pulse oximeter readings to adjust for the decrease in atmospheric pressure and oxygen concentration at higher elevations.
Therefore, when selecting a pulse oximeter for use at higher elevations, it is essential to choose one that has been validated and calibrated for use at these altitudes. Several manufacturers produce pulse oximeters with an altitude adjustment feature, which corrects for altitude-induced hypoxemia. For example, the Nonin 9560 Onyx Vantage Finger Pulse Oximeter has an altitude compensation range of up to 12,000 feet and has been tested for accuracy at high altitudes (Nonin Medical Inc, 202451).
Arterial oxygen saturation (SPo2), Early hypoxemia, a decrease in the Spo2 greater than that expected for a given altitude, is a risk factor for AMS (Burtscher et al., 200452; Karinen et al. 201053). An individual with early hypoxemia needs to be urged to avoid intense activity and, if continuing to ascend, to do so gradually. In order to monitor Spo2 persons with deteriorating AMS, HACE, and HAPE symptoms, trekking firms frequently carry pulse oximeters, which are reasonably priced with the aid of a pulse oximeter, early hypoxemia can be observed (Figure 2).

This figure shows a Nonin portable pulse oximeter used to measure oxygen saturation (%SpO₂) and heart rate. The device is red, small, and easy to carry, making it suitable for use in the field. The display shows an oxygen saturation reading of 98% and a heart rate of 77 beats per minute. A green light at the bottom indicates the device is working properly. This oximeter was used during the study for accurate and non-invasive data collection.
This image illustrates the working principle of a pulse oximeter. It shows a cross-section of a finger placed between two parts of the device. The top component emits red and infrared light, which passes through the finger, while the bottom part detects the light. The red and blue lines inside the finger represent arteries and veins, highlighting blood flow. This process measures oxygen saturation by analyzing how light is absorbed by oxygenated and deoxygenated hemoglobin. The non-invasive method allows quick and accurate monitoring of blood oxygen levels.
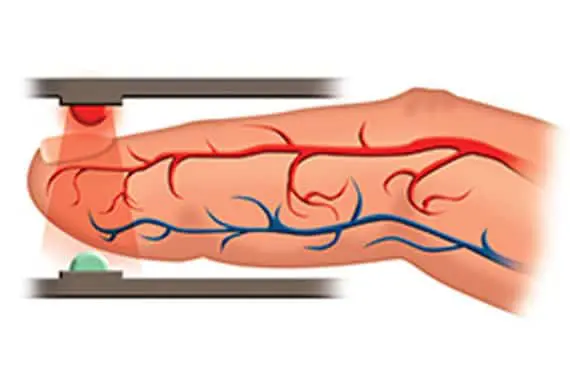
Figure 2: Pulse oximeter and its mechanism
Research Method and Methodology
This study adopted the abductive approach based on statistical, analytical, and experimental field-based methods. Data will be gathered through both primary and secondary sources. Primary field observation was made during the expedition to Tsergo Ri (~4990 asl) and a secondary review of international practice of medical journals, articles, and medical websites. The findings were discussed based on primary fieldwork data and a literature review. The report has attempted to study the “Impact of Smoking Habit, Gender, and Altitude on High Altitude Illness: A Comparative Study in the Langtang Region of Nepal” with the help of medical parameters (vital signs); Pulse (heat rate in bpm), Arterial oxygen saturation Spo2 in % and High-altitude illness (AMS) evaluated by Lake Louise Score (no symptoms-0 to extreme symptoms-3) (Roach et al., 201854). The research used a mixture of social science and pure science tools. Social science tools such as interviewing, questionnaires, and observation, and pure science tools are based on the scientific interpretation of the researcher.
Study Area
The study area is situated in the Langtang region, Rasuwa district, Gosaikunda Municipality, specifically at Tsergo Ri Peak.
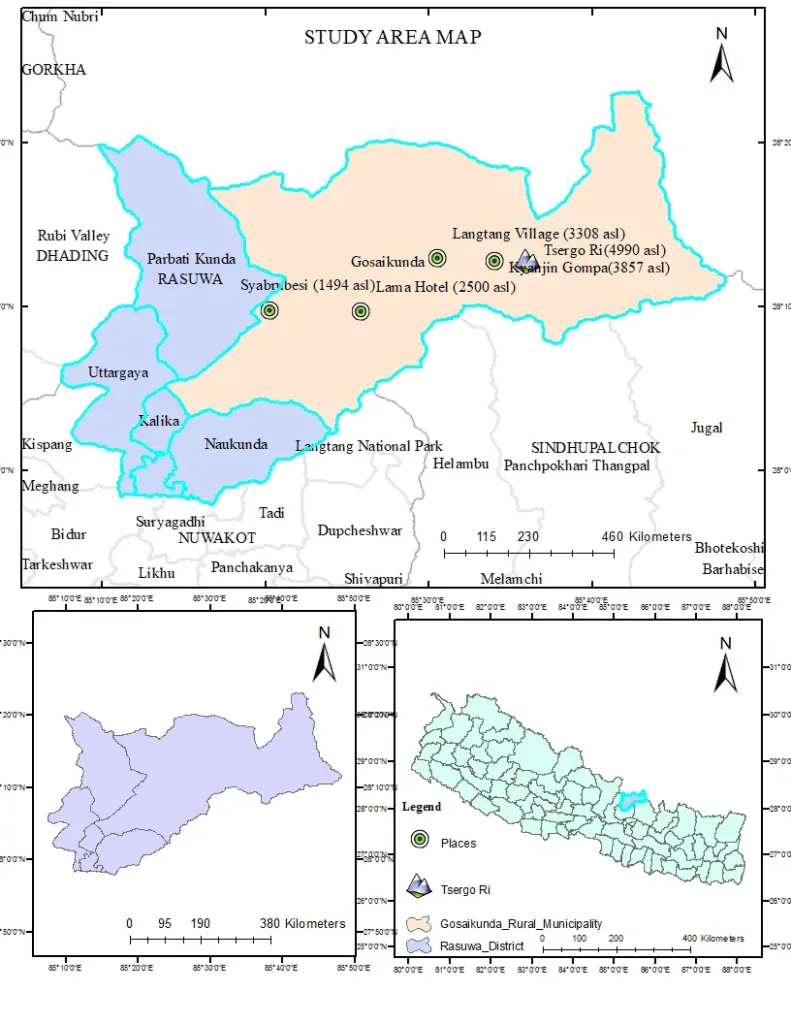
Methods and Methodology
A total of 33 individuals participated in the study. All participants were students from the third batch of Nepal Mountain Academy (NMA) and included one IFMGA guide, a field supervisor, and an assistant supervisor. Most participants had not been exposed to high altitude within six months prior to the study, except for the IFMGA guide and five other subjects. The physical fitness of participants was assessed using the acute mountain sickness (AMS) score, as per the Lake Louise agreement (no symptoms = 0; extreme symptoms = 2 or 3) (Roach et al., 201855).
All data were expressed as mean ± standard deviation (SD), median, variance, skewness, kurtosis, and range. Statistical comparisons of these values were performed using analysis of variance with frequency. Independent samples Mann-Whitney U tests were conducted to compare SpO₂ and heart rate. Paired samples tests were used to compare SpO₂ and heart rate measurements between Langtang Village (LV) and Tsergo Ri (TR) at two different elevations. These analyses were performed using IBM Statistical Package for the Social Sciences (SPSS) software.
Data Processing and Analysis
Data processing and analysis were conducted after the trek using IBM Statistical Package for the Social Sciences (SPSS) software. Following the heart rate variability and SpO₂ measurements, participants were evaluated using the Lake Louise Scoring (LLS) system for acute mountain sickness (AMS). The LLS system includes a self-report questionnaire assessing the presence and severity of symptoms and a clinical assessment (Hackett & Oelz, 1992). The total LLS score was calculated by combining the clinical assessment score with the self-reported questionnaire score, and AMS was diagnosed accordingly.
Statically Analysis
Continues values were presented as Mean, Median, standard deviation (SD), Variance, Skewness, Kurtosis, and range. Statistical comparison of Mean, Median, standard deviation (SD), Variance, Skewness, Kurtosis, and range. The frequency of parameters had assessed by Paired samples tests which conducted to compare the Spo2 and heart rate measurements between Langtang Village (LV) and Tsergo RI (TR) at two different elevations. The sample consisted of 33 participants who provided measurements at both locations.
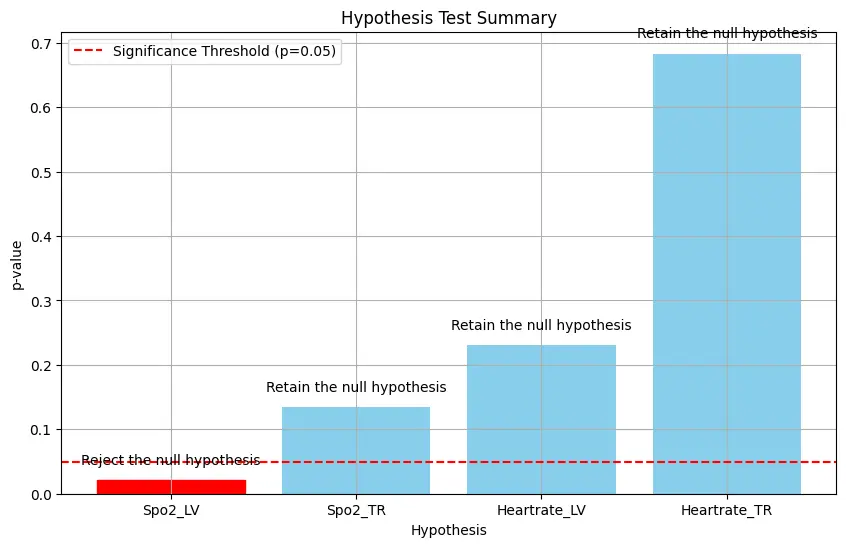
Independent samples Mann-Whitney U tests were conducted to compare Spo2 and heart rate between genders separately for Langtang Village (LV) and Tsergo RI (TR). The significance level was set at α = 0.05.
Presentations and Tools
Tabulation, pictorial and graphical method of analysis and interpretation were presented the data. Simultaneously, Tabulation of data helps too researcher or onlooker that helps to take grasp of data and comprehend the idea and immediately found that what he/she looking for. Data analysis and presentation is done with the following tools;
a. Oximeter (Pulse Oximeter):Entire the study area with different high altitude using Pulse Oximeter for taking the Pulse (heart rate in bmp unit) and Oxygen Saturation (SPO₂ in %).
b. SPSS: SPSS, Statistical Package for the social science is a Statistical software suite developed by IBM for data management that is used for analysis, interpretation and presentation of data in different form like graph and table.
c. ArcGIS: ArcGIS, used for make study area is quite useful software for explore and create the unexplored routs and maps.
Results and Discussions
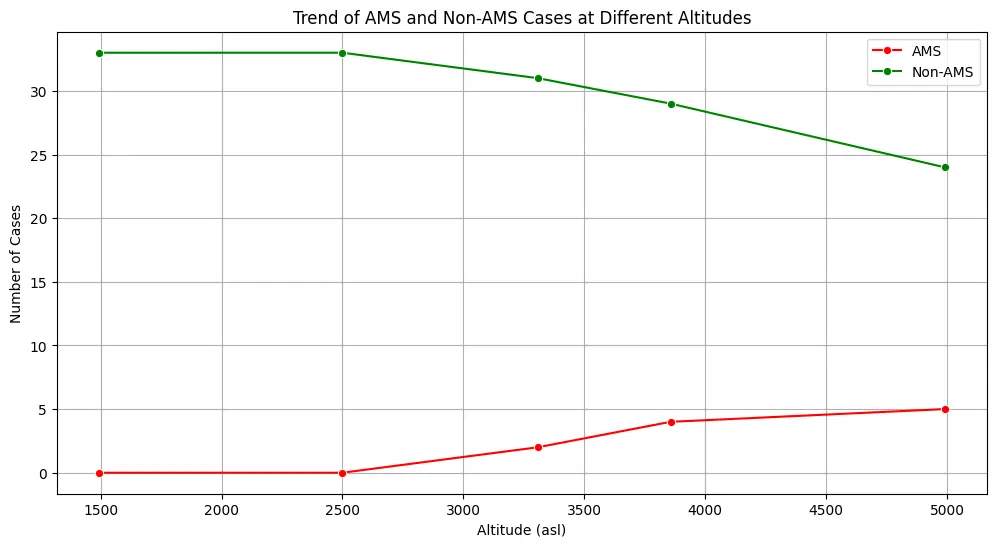
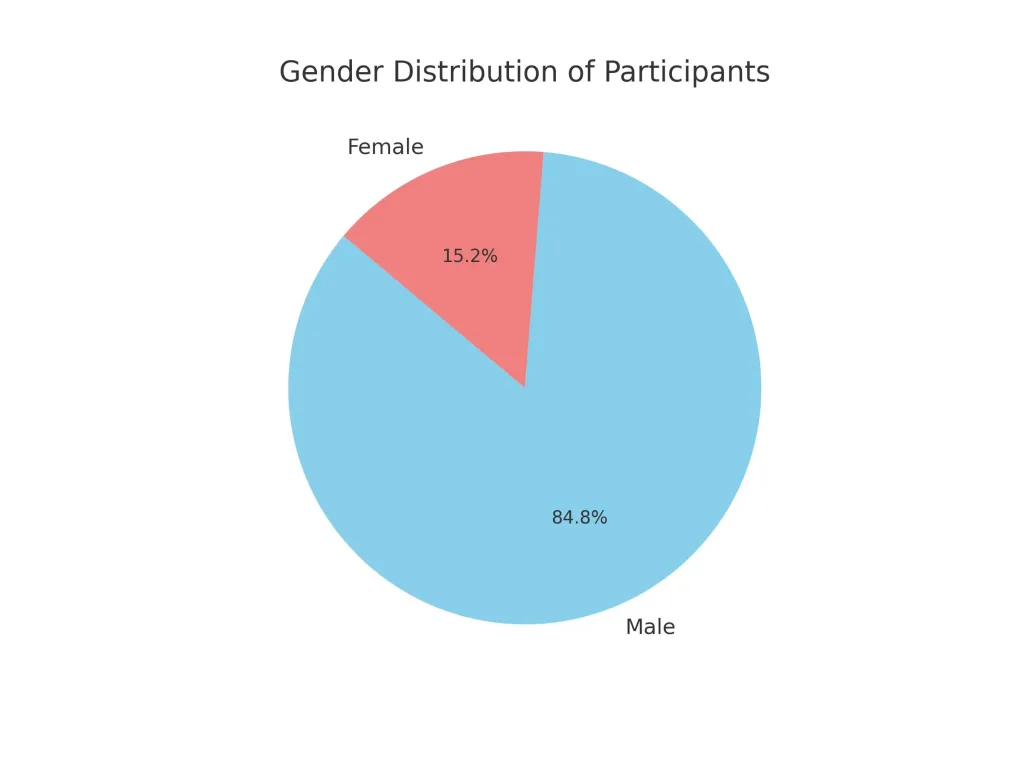
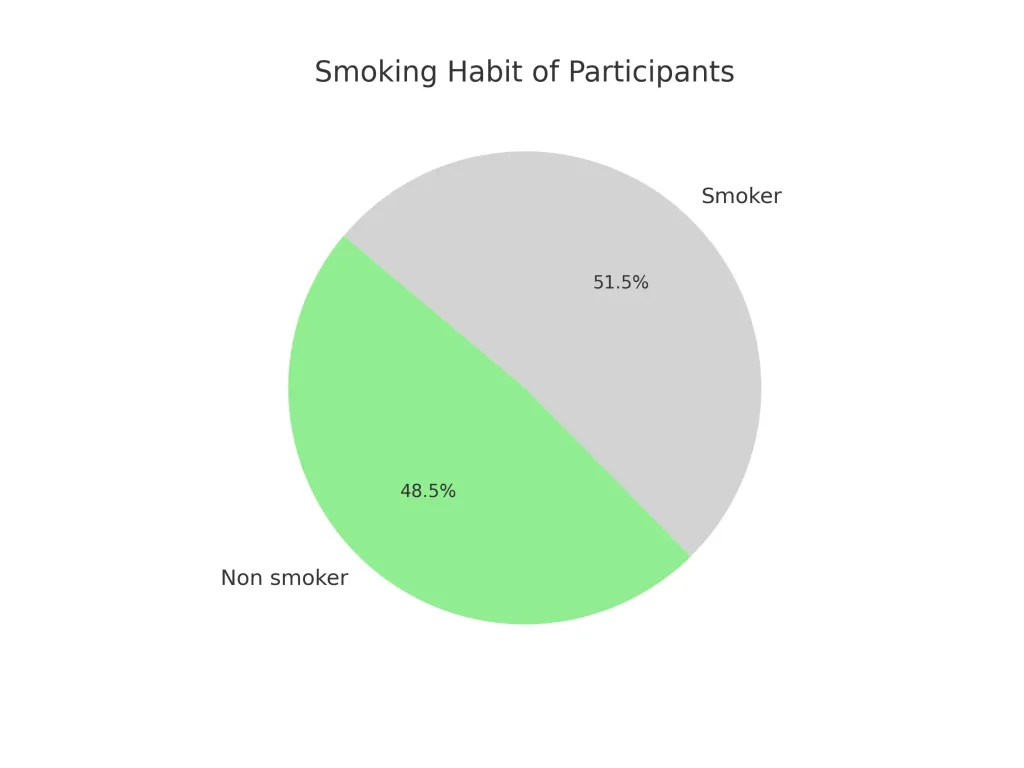
The study involved 33 participants, with a demographic distribution of 88.8% males (n=28) and 15.2% females (n=5) at different altitudes (figure 4). Smoking habits were noted, revailing 48.5% non smokers (n=16) and 51.5% smokers (n=17), with all female participants being non-smokers.
The Spo2 and heart rate measurements were recorded at low altitudes (Langtang Village) and High altitudes (Tsergo Ri). At LV, the mean SP02 was 88.758% (SD=3.4644), decreasing to 78.517% (SD=6.2714) at TR. The mean heart rate increased from 92.030 bpm (SD=13.2015) at LV to 111.241 bpm (SD=15.4265) at TR. These finding are consistent with the physiological responses reported by west (201256) and Richalet et al., (201257), who observed decreased SPO2 and increased heart rate at higher altitudes due to reduced oxugen availability and compensatory mechanisms.
Further analysis comparing smokers and non smokers indicated that smokers had a lower mean SPO2 at both LV (87.353%) and TR (76.643%), with higher heart rates at LV (94.353 bpm) and TR (109.571 bpm). Non Smokers exhibited higher SPO2 levels at LV (90.250%) and TR (80.267%), with slightly lower heart rate at LV (89.563 bpm) and TR (112.800bpm). These results align with Basnyat and Murdoch (200358), who found that smoking exacerbates hypoxemia and increases heart rates at high altitudes due to addiational oxidative stress and impaired oxygen transport.
Gender differences were also examined, showing that female had higher mean SPO2 at both LV (90.800%) and TR (78.000%) compared to males (88.393% at LV and 78.600% at TR). Heart rate were also higher for Females at LV (101.00 bpm) but slightly lower at TR (107.500 bpm) compared to males (90.429 bpm at LV and 111.840 bpm at TR). Although not stastically significant, these differences suggest potential gender-related physiological adapatations to altitude, supporting findings by Richalet et al. (201259).
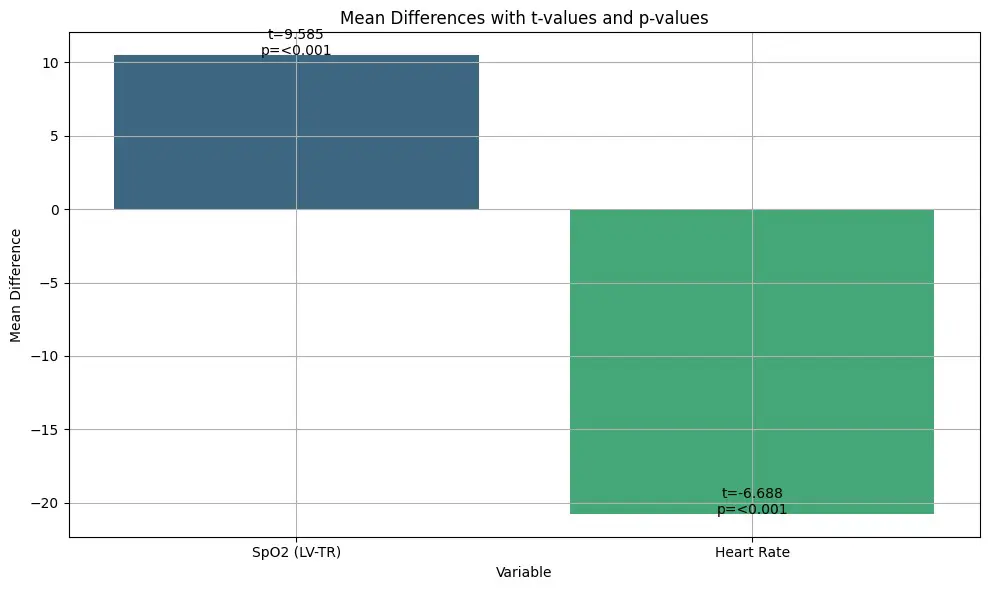
Stastical analyses, including paired sample t-tests and Mann-Whitney U tests, confirmed significant differences in SPO2 (p<0.001) and heart rate (p<0.001) between LV and TR, affirming the expected physiological response to higher altitudes with significant decreases in SPO2 and an increase in heart rate. These findings are consistent with the established scientific understanding that reduced oxygen availability at higher altitudes leads to hypoxemia and a compensatory increase in heart rate (west, 201260). Addiationally, Mann-Whitney U tests highlighted significant differences in SPO2 distribution among smokers and LV (p=0.013), consistent with the literature on the compounded effects of smoking and altitude (Basnyat & Murdoch, 200361).
West (201262) and Richalet et al., (201263) discuss the physiological mechanisms behind the decrease in SPO2 and increase in heart rate at higher altitudes, highlighting that hypoxemia triggers increased ventilation and cardic output to enhance oxygen delivery. The impact of smoking on altitude adaptation has been extensively studied, with Basnet and Murdoch (200364) highlighting the greater challenges smokers face in maintaining adequate oxygenation due to impaired lung function and increased oxidative stress. Gender differences in altitude adapatation are less clear-cut. Still, Richalet et al. (201265) suggest that women might have an adavantage in maintaining oxygen saturation, possible due to differences in haemoglobin levels and harmonal influences. Our Study observed higher SPO2 levels in females at both altitudes, indicating the need for further research in this area.
The result show a clear pattern of decreasing SPO2 and increasing heart rate with rising altitude, reflecting the physiological adjustments necessary to cope with hypoxic conditions. Smokers consistently showed lower SPO2 and higher heart rates compared to non-smokers, emphasising the detrimental impact if smoking on respiratory and cardiovascular function at high altitudes. Gender differences, though not stastically significant, suggest that females might have a slight advantage in maintaining oxygen saturation levels, aligning with some reports in the lioterature suggesting better acclimatization in women (Richalet et al., 201266).
Conclusion
The Study provides valuable insight into the effects of altitude, gender, and smoking on arterial oxygen saturation (SPO2%) and heart rate. As altitude increased from Langtang Village (LV) to tsergo Ri (TR), average SPO2% level in smokers decreased from 87.35% to 76.64%, while heart rates increased from 95.52 bpm to 111.24 bpm. Smokers consistently exhibited lower SPO2 levels and higher heart rate than non-smokers. Gender defferences were also observed, with females generally displaying higher SPO2 % values than males at both males at both altitudes. For instance, at LV, females had an avarage heart rate of 101 bpm compared to 90.43 bpm for males.
Based on a sample of 33 participants (84.8 % nmale, 15.2 % females; 51.5% smokers, 48.5 % non -smokers), these findings underscore the importance of understanding physiological changes at high altitudes and recognising the addiational risks posed by smoking in these environments. The study’s results can inform safety protocols and healt education initiatives for individuals exposed to high altitude conditions. Howevcr, further research with more extensive and diverse samples is recommended to explore these factors and their interactions further.
References
- Saito, S., Tanobe, K., Yamada, M., & Nishihara, F. (2005). Relationship between arterial oxygen saturation and heart rate variability at high altitudes. American journal of Emergency Medicine, 23(1), 8-12. https://soi.org/10.1016/j.ajem.2004.09.023 ↩︎
- Moghaddasi, B. (2019). High Altitude illness in Annapurna circuit Trek. Journal of Tourism and Himalayan Adventure, 1(1), 14-27. ↩︎
- Wall, I., & Paudel, T.P. (2021). Adventure and mountaineering Tourism. Kathmandu: Nepal Mountain Academy ↩︎
- Basnyat, B., & Murdoch, D. R. (2003). High-altitude illness. Lancet, 361(9373), 1967–1974. https://doi.org/10.1016/s0140-6736(03)13591-x ↩︎
- Karinen, H. M., Peltonen, J. E., Kahonen, M., & Tikkanen, H. O. (2010). Prediction of acute mountain sickness by monitoring arterial oxygen saturation during ascent. High Altitude Medicine and biology, 11 (4), 325-332. https://doi.org/10.1089/ham.2009.1060 ↩︎
- Karinen, H., Peltonen, J., & Tikkanen, H. (2008). Prevalence of acute mountain sickness among finish trekkers on Mount Kilimanjaro, Tanzania: An observatioanal study. High Altitude Medicine and Biology, 9(4), 301-306. https://doi.org/10.1089/ham.2008.1008 ↩︎
- Luks, A. M., Swenson, E. R., & Bärtsch, P. (2017). Acute high-altitude sickness. European Respiratory Review, 26(143), 160096. https://doi.org/10.1183/16000617.0096-2016 ↩︎
- Moghaddasi, B. (2019). High Altitude illness in Annapurna circuit Trek. Journal of Tourism and Himalayan Adventure, 1(1), 14-27. https://nma.gov.np/storage/listies/November2020/journal-of-nepal-mountain-academy-2019-1st-issue.pdf ↩︎
- Saito, S., Tanobe, K., Yamada, M., & Nishihara, F. (2005). Relationship between arterial oxygen saturation and heart rate variability at high altitudes. American journal of Emergency Medicine, 23(1), 8-12. https://soi.org/10.1016/j.ajem.2004.09.023 ↩︎
- Eter, P., Ackett, H.H., Obert, R., & Oach, C.R. (2001). High altitude illness. New England Journal of Medicine, 345, 107-114. DOI: 10.1056/NEJM200107123450206 ↩︎
- Eter, P., Ackett, H.H., Obert, R., & Oach, C.R. (2001). High altitude illness. New England Journal of Medicine, 345, 107-114. DOI: 10.1056/NEJM200107123450206 ↩︎
- Karinen, H., Peltonen, J., & Tikkanen, H. (2008). Prevalence of acute mountain sickness among finish trekkers on Mount Kilimanjaro, Tanzania: An observatioanal study. High Altitude Medicine and Biology, 9(4), 301-306. https://doi.org/10.1089/ham.2008.1008 ↩︎
- Hackett, P.H., Rennie, D., & Levine, H.D. (1976). The incidence, importance, and prophylaxis of acute mountain sickness. The Lancet. 308(7996). 1149-1155. https://doi.org/10.1016/S0140-6736(76)91677-9 ↩︎
- Meier, D., Collet, T.H., Locatelli, I., Cornuz, J., kaiser, B., Simel, D.L., & Sartori, C. (2017). Does this patient have acute mountain sickness? The rational clinical examination systematic review. Journal of the American medical Association, 318(18), 1810-1819. https://doi.org/10.1001/jama.2017.16192 ↩︎
- Richalet, J., Larmignat, P., Poitrine, E., Letournel, M., & Canouï-Poitrine, F. (2011). Physiological risk factors for severe High-Altitude illness. American Journal of Respiratory and Critical Care Medicine, 185(2), 192–198. https://doi.org/10.1164/rccm.201108-1396oc ↩︎
- Wall, I., & Paudel, T.P. (2021). Adventure and mountaineering Tourism. Kathmandu: Nepal Mountain Academy ↩︎
- Gallagher, S. A., & Hackett, P.H. (2004). High-altitude illness. Emergency Medicine Clinics of North America. 41(2). 329-355. https://doi.org/10.1016/j.emc.2004.02.001 ↩︎
- Gallagher, S. A., & Hackett, P.H. (2004). High-altitude illness. Emergency Medicine Clinics of North America. 41(2). 329-355. https://doi.org/10.1016/j.emc.2004.02.001 ↩︎
- Gallagher, S. A., & Hackett, P.H. (2004). High-altitude illness. Emergency Medicine Clinics of North America. 41(2). 329-355. https://doi.org/10.1016/j.emc.2004.02.001 ↩︎
- Wall, I., & Paudel, T.P. (2021). Adventure and mountaineering Tourism. Kathmandu: Nepal Mountain Academy ↩︎
- Hevroni, A., Goldman, A., & kerem, E. (2015). High altitude physiology and pathophysiology in adults and children: A review. Clinical Pulmonary medicine, 22(3), 105-113. https://doi.org/10.1097/CPM.0000000000000093 ↩︎
- Eter, P., Ackett, H.H., Obert, R., & Oach, C.R. (2001). High altitude illness. New England Journal of Medicine, 345, 107-114. DOI: 10.1056/NEJM200107123450206 ↩︎
- Kurtzman, R. A., & Caruso, J.L. (2018). High Altitude Illness Death investigation. IN Academic Forensic Pathology 8(1) 83-97. https://doi.org/10.23907/2018.006 ↩︎
- Roach, R. C., Hackett, P. H., Oelz O., Bartsch, P., Luks, A.M., Maclnnis, M.J., Baillie, J. K., Achatz, E., Albert, E., Zafren, K., Yaron, M. Willmann, G., wilkes, M., West, J.B., Wang, S.H., Wagner, D.R., Voituron, N., Ulrich, S., Twomey, R., & Andrews, J.S. (2018). The 2018 lake louise acute mountain sickness score. High Altitude Medicine and Biology, 19(1), 4-6. https://doi.org/10.1089/ham.2017.0164 ↩︎
- Moghaddasi, B. (2019). High Altitude illness in Annapurna circuit Trek. Journal of Tourism and Himalayan Adventure, 1(1), 14-27. ↩︎
- Wall, I., & Paudel, T.P. (2021). Adventure and mountaineering Tourism. Kathmandu: Nepal Mountain Academy ↩︎
- Basnyat, B., & Murdoch, D. R. (2003). High-altitude illness. Lancet, 361(9373), 1967–1974. https://doi.org/10.1016/s0140-6736(03)13591-x ↩︎
- Karinen, H. M., Peltonen, J. E., Kahonen, M., & Tikkanen, H. O. (2010). Prediction of acute mountain sickness by monitoring arterial oxygen saturation during ascent. High Altitude Medicine and biology, 11 (4), 325-332. https://doi.org/10.1089/ham.2009.1060 ↩︎
- Maggiorini, M., MéLot, C., Pierre, S., Pfeiffer, F., Greve, I., Sartori, C., Lepori, M., Hauser, M., Scherrer, U., & Naeije, R. (2001). High-Altitude pulmonary edema is initially caused by an increase in capillary pressure. Circulation, 103(16), 2078–2083. https://doi.org/10.1161/01.cir.103.16.2078 ↩︎
- Roach, R. C., Hackett, P. H., Oelz O., Bartsch, P., Luks, A.M., Maclnnis, M.J., Baillie, J. K., Achatz, E., Albert, E., Zafren, K., Yaron, M. Willmann, G., wilkes, M., West, J.B., Wang, S.H., Wagner, D.R., Voituron, N., Ulrich, S., Twomey, R., & Andrews, J.S. (2018). The 2018 lake louise acute mountain sickness score. High Altitude Medicine and Biology, 19(1), 4-6. https://doi.org/10.1089/ham.2017.0164 ↩︎
- Roach, R.C., Maes, D., Sandoval, D., Robergs, R.A., Icenogle, M., Hinghofer-Szalkay, H., Lium, D., & Loeppky, J. A. (2000). Exercise exacerbates acute mountain sickness at simulated high altitude. Journal of Applied Physiology, 88(2), 581-585. https://doi.org/10.1152/jappl.2000.88.2.581 ↩︎
- Moghaddasi, B. (2019). High Altitude illness in Annapurna circuit Trek. Journal of Tourism and Himalayan Adventure, 1(1), 14-27. ↩︎
- Gallagher, S. A., & Hackett, P.H. (2004). High-altitude illness. Emergency Medicine Clinics of North America. 41(2). 329-355. https://doi.org/10.1016/j.emc.2004.02.001 ↩︎
- Fulco, C. S., Rock, P. B., & Cymerman, A. (1998). Maximal and submaximal exercise performance at altitude. PubMed, 69(8), 793–801. https://pubmed.ncbi.nlm.nih.gov/9715971 ↩︎
- Society, W. M., & Forgey, W. W. (1995). Wilderness Medical Society Practice guidelines for wilderness Emergency care. Globe Pequot Press. ↩︎
- Fulco, C. S., Rock, P. B., & Cymerman, A. (2000). Improving athletic performance: is altitude residence or altitude training helpful? PubMed, 71(2), 162–171. https://pubmed.ncbi.nlm.nih.gov/10685591 ↩︎
- Banderet, L. E., & Shukitt-Hale, B. (2002). Cognitive performance, mood, and neurological status at high terrestrial elevation [Dataset]. In PsycEXTRA Dataset. https://doi.org/10.1037/e447232006-001 ↩︎
- Society, W. M., & Forgey, W. W. (1995). Wilderness Medical Society Practice guidelines for wilderness Emergency care. Globe Pequot Press. ↩︎
- Hongiman, B., Kay Theis, M., Koziol-McLain, J., Roach, R., Yip, R., Houston, C., & moore, L.G. (1993). Acute Mountain Sickness in a General Tourist Population at Moderate Altitudes. Annals Of Internal Medicine, 118(8), 587-592. https://doi.org/10.7326/0003-4819-118-8-199304150-00003 ↩︎
- Bisgard, G. E., & Forster, H. V. (1996). Ventilatory responses to acute and chronic hypoxia. Comprehensive Physiology, 1207–1239. https://doi.org/10.1002/cphy.cp040252 ↩︎
- Durkot, M., Hoyt, R., Darrigrand, A., Hubbard, L., Kamimori, G., & Cymerman, A. (1996). Chronic hypobaric hypoxia decreases intracellular and total body water in microswine. Comparative Biochemistry and Physiology Part a Physiology, 114(2), 117–121. https://doi.org/10.1016/0300-9629(95)02103-5 ↩︎
- Sawka, M. N., Convertino, V. A., Eichner, E. R., Schnieder, S. M., & Young, A. J. (2000). Blood volume: importance and adaptations to exercise training, environmental stresses, and trauma/sickness. Medicine & Science in Sports & Exercise, 32(2), 332. https://doi.org/10.1097/00005768-200002000-00012 ↩︎
- Rs, K., & Sm, C. (1967). Effects of acetazolamide on physiologic and subjective responses of men to 14,000 feet. SAM-TR-67-81. PubMed, 1–10. https://pubmed.ncbi.nlm.nih.gov/5302807 ↩︎
- Beidleman, B. A., Muza, S. R., Fulco, C. S., Cymerman, A., Ditzler, D., Stulz, D., Staab, J. E., Skrinar, G. S., Lewis, S. F., & Sawka, M. N. (2004). Intermittent altitude exposures reduce acute mountain sickness at 4300 m. Clinical Science, 106(3), 321–328. https://doi.org/10.1042/cs20030161 ↩︎
- Eter, P., Ackett, H.H., Obert, R., & Oach, C.R. (2001). High altitude illness. New England Journal of Medicine, 345, 107-114. DOI: 10.1056/NEJM200107123450206 ↩︎
- Society, W. M., & Forgey, W. W. (1995). Wilderness Medical Society Practice guidelines for wilderness Emergency care. Globe Pequot Press. ↩︎
- Moghaddasi, B. (2019). High Altitude illness in Annapurna circuit Trek. Journal of Tourism and Himalayan Adventure, 1(1), 14-27. ↩︎
- Roach, R.C., Maes, D., Sandoval, D., Robergs, R.A., Icenogle, M., Hinghofer-Szalkay, H., Lium, D., & Loeppky, J. A. (2000). Exercise exacerbates acute mountain sickness at simulated high altitude. Journal of Applied Physiology, 88(2), 581-585. https://doi.org/10.1152/jappl.2000.88.2.581 ↩︎
- Moghaddasi, B. (2019). High Altitude illness in Annapurna circuit Trek. Journal of Tourism and Himalayan Adventure, 1(1), 14-27. ↩︎
- Robergs, R. A., & Landwehr, R. (2002). The surprising history of the “HRmax=220-age” equation. Journal of Exercise Physiology, 5(2), 1–16. https://eprints.qut.edu.au/96880/ ↩︎
- Nonin Medical Inc. (2024, December 2). Pulse and regional oximeters, capnographs, sensors | Nonin. Nonin. https://www.nonin.com/ ↩︎
- Burtscher, M., Pachinger, O., Ehrenbourg, I., Mitterbauer, G., Faulhaber, M., Pühringer, R., & Tkatchouk, E. (2004). Intermittent hypoxia increases exercise tolerance in elderly men with and without coronary artery disease. International Journal of Cardiology, 96(2), 247–254. https://doi.org/10.1016/j.ijcard.2003.07.021 ↩︎
- Karinen, H. M., Peltonen, J. E., Kähönen, M., & Tikkanen, H. O. (2010). Prediction of acute mountain sickness by monitoring arterial oxygen saturation during ascent. High Altitude Medicine & Biology, 11(4), 325–332. https://doi.org/10.1089/ham.2009.1060 ↩︎
- Roach, R. C., Hackett, P. H., Oelz, O., Bärtsch, P., Luks, A. M., MacInnis, M. J., Baillie, J. K., Achatz, E., Albert, E., Andrews, J. S., Anholm, J. D., Ashraf, M. Z., Auerbach, P., Basnyat, B., Beidleman, B. A., Berendsen, R., Berger, M. M., Bloch, K. E., Brugger, H., . . . Zafren, K. (2018). The 2018 Lake Louise Acute Mountain Sickness score. High Altitude Medicine & Biology, 19(1), 4–6. https://doi.org/10.1089/ham.2017.0164 ↩︎
- Roach, R. C., Hackett, P. H., Oelz, O., Bärtsch, P., Luks, A. M., MacInnis, M. J., Baillie, J. K., Achatz, E., Albert, E., Andrews, J. S., Anholm, J. D., Ashraf, M. Z., Auerbach, P., Basnyat, B., Beidleman, B. A., Berendsen, R., Berger, M. M., Bloch, K. E., Brugger, H., . . . Zafren, K. (2018). The 2018 Lake Louise Acute Mountain Sickness score. High Altitude Medicine & Biology, 19(1), 4–6. https://doi.org/10.1089/ham.2017.0164 ↩︎
- West, B.J. (2012). Fractal Physiology and Chos in Medicine: 2nd Edition. https://www.amazon.com/fracral-physiology-medicine-Nonlinear-Phenomena/dp/9814417793 ↩︎
- Richalet, J., Larmignat, P., poitrine, E., Letournel, M., & Canoui-Poitrine, F. (2012). Physiology risk factors for severe High-Altitude illness. American Journal of respiratory and critical care medicine, 185 (2). 192-198. https://doi.org/10.1164/rccm.201108-1396oc ↩︎
- Basnyat, B., & Murdoch, D. R. (2003). High-altitude illness. lancet, 361(9373), 167-1974. https://doi.org/10.1016/s0140-6736(03)13591-x ↩︎
- Richalet, J., Larmignat, P., poitrine, E., Letournel, M., & Canoui-Poitrine, F. (2012). Physiology risk factors for severe High-Altitude illness. American Journal of respiratory and critical care medicine, 185 (2). 192-198. https://doi.org/10.1164/rccm.201108-1396oc ↩︎
- West, B.J. (2012). Fractal Physiology and Chos in Medicine: 2nd Edition. https://www.amazon.com/fracral-physiology-medicine-Nonlinear-Phenomena/dp/9814417793 ↩︎
- Basnyat, B., & Murdoch, D. R. (2003). High-altitude illness. lancet, 361(9373), 167-1974. https://doi.org/10.1016/s0140-6736(03)13591-x ↩︎
- West, B.J. (2012). Fractal Physiology and Chos in Medicine: 2nd Edition. https://www.amazon.com/fracral-physiology-medicine-Nonlinear-Phenomena/dp/9814417793 ↩︎
- Richalet, J., Larmignat, P., poitrine, E., Letournel, M., & Canoui-Poitrine, F. (2012). Physiology risk factors for severe High-Altitude illness. American Journal of respiratory and critical care medicine, 185 (2). 192-198. https://doi.org/10.1164/rccm.201108-1396oc ↩︎
- Basnyat, B., & Murdoch, D. R. (2003). High-altitude illness. lancet, 361(9373), 167-1974. https://doi.org/10.1016/s0140-6736(03)13591-x ↩︎
- Richalet, J., Larmignat, P., poitrine, E., Letournel, M., & Canoui-Poitrine, F. (2012). Physiology risk factors for severe High-Altitude illness. American Journal of respiratory and critical care medicine, 185 (2). 192-198. https://doi.org/10.1164/rccm.201108-1396oc ↩︎
- Richalet, J., Larmignat, P., poitrine, E., Letournel, M., & Canoui-Poitrine, F. (2012). Physiology risk factors for severe High-Altitude illness. American Journal of respiratory and critical care medicine, 185 (2). 192-198. https://doi.org/10.1164/rccm.201108-1396oc ↩︎